- Home
- About
- Contact
- Driverpack solution 16 full
- Download camtasia studio 8
- Autodesk maya 2014 bittorrent
- Charles bronson last photo
- Expert advisor generator
- Hasbi rabbi jallallah mp3 free download
- Soap tv show full episodes
- 70-2087 motionpro
- Best starcraft 2 game
- Anime studio pro 11-2
- Download lagu iwan fals ibuku
- Waves vocal rider review
- Adobe premiere pro cs4 troubleshooting
- Asterix and cleopatra poster
- Alan walker on my way espa-ol
- Usb endoscope focus
- Empire season 2 episode 1 andre
- Saving troikatronix isadora files to older versions
- Anthropics portrait professional studio 12
- How to crack sony vegas movie studio hd platinum 11
- Leica geo office 3-0
- What photoshop do i use with brother gt 541
- Tv for sale
- Chemical equation balancer claculator

Major areas of applications are process industries and energy production, such as oil refining, natural gas processing, petrochemistry, chemical industries, mineral processing and utility production and distribution systems.
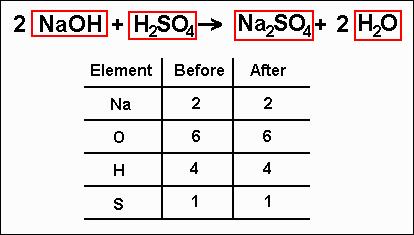
The book is directed to chemical engineers, plant designers, technologists, information technology managers, control engineers and instrumentation engineers in process industries. For example, your equation should look something like 'H2 + O2 H2O.' Count the number of atoms in each element on each side of the equation and list them under that side. But I think well see that if we work through this carefully and methodically, and we also appreciate the art of balancing chemical equations, that its actually not too bad. To balance a chemical equation, first write out your given formula with the reactants on the left of the arrow and the products on the right. The answer will appear below Always use the upper case for the first character in the element name and the lower case for the second character. The major areas addressed are: - single- and multi-component balancing - energy balance - entropy and exergy (availability) balances - solvability of balancing problems - balancing with data reconciliation - dynamic balancing - measurement design and optimisation - regular balancing of large industrial systems. Balancing chemical equations is one of those concepts in chemistry that often confuses people. Direct link to this balanced equation: Instructions on balancing chemical equations: Enter an equation of a chemical reaction and click 'Balance'. The classical treatment of balances in the available literature is complemented in the following areas: - systematic analysis of large systems by Graph theory - comprehensive thermodynamic analysis (entropy and availability) - balancing on the basis of measured plant data (data reconciliation) - measurement design and optimisation - dynamic balancing - plant-wide regular mass and energy balancing as a part of company's information system. Balancing chemical equation is the process of equalising the number of each Nomenclature, a collection of rules for naming things, is important in science. There is also another Oxygen, but it is in a different molecule so you need to add it to the 14 to get a total of 15 atoms for Oxygen.ģ.This book represents the systematic coverage of mass and energy balancing in the process industries. If, for example, you write 2 H 2 O, that means you have 2 times the number of atoms in each water molecule, which would be 4 hydrogen atoms and 2 oxygen atoms. Coefficients are whole number multipliers. When balancing equations, you never change subscripts. Since the Carbon molecule also has oxygen in it, you need to multiply the 7 by the number of atoms with Oxygen which is 2 to get a total of 14. Add Coefficients To Balance Mass in a Chemical Equation. Edit Mode lets you enter or edit the chemical equation and Balance Mode. Since you had 7 Carbons on the left and 1 Carbon on the right, you are going to need to have a 7 as the coefficient on the right.Ĭ. This tool can be used alone, or you can copy/paste it into another TNS document. Sometimes you get an odd number of oxygens on the right which leads to a fraction in. The greatest common divisor and the lowest common multiple of two integers. It is balanced Actually, it doesnt always work out that easily. Adjust the table by multiplying each element effected by the coefficient added to that molecule.ī. Gaussian Method for system of linear equations with any number of variables. Once you've determined which element to balance first, add a coefficient to balance that element by multiplying the subscript by the coefficient added.Ī. For a more complicated example that has more than one element besides Oxygen or Hydrogen, pick the element that has more atoms on one side compared to the other.Ģ.

Leave hydrogen and oxygen for last because they are more often found in more than one chemical on each side of the equation.ī. Chemical reaction: process in which one or more substances, the reactants, are converted to one or more different substances, the products. Pick an element that appears in one molecule on the left side and in one molecule on the left.Ī. Description: Flash animation with 3 chemical reactions to balance (find the stoichiometric coefficients).
